The immunisation, currently being developed by researchers at USP, is based on a technology known as “virus-like particles” (VLPs), which does not use the pathogen’s genetic material

 A new Zika virus vaccine developed by USP researchers has been shown to be safe and effective in mouse trials. In addition to inducing the immune response against the pathogen, the immunisation also protected the animals from brain and testicular damage associated with viral infection. The findings were published in the scientific journal NPJ Vaccines and represent an important step in Zika prevention strategies.
A new Zika virus vaccine developed by USP researchers has been shown to be safe and effective in mouse trials. In addition to inducing the immune response against the pathogen, the immunisation also protected the animals from brain and testicular damage associated with viral infection. The findings were published in the scientific journal NPJ Vaccines and represent an important step in Zika prevention strategies.
“It is ten years since the Zika epidemic in Brazil and the disease continues to be a threat to public health, especially for pregnant women and their babies. In the study, we were able to design a formulation capable of neutralizing the pathogen and protecting rodents from both inflammation in the brain – one of the most worrying consequences of infection – and testicular damage, something that has not been observed in epidemiological studies, but is a hallmark of the disease when studied in the laboratory”, says Gustavo Cabral Miranda, a collaborating researcher at USP’s Institute of Biomedical Sciences (ICB) and responsible for the project.
Miranda explains that the strategy used in the formulation is based on a technology known as “virus-like particles” (VLPs). “Unlike more traditional strategies, which use attenuated or inactivated virus inoculation, in this formulation we do not use the genetic material of the pathogen, which makes its development much safer and more economical, and eliminates the need for substances that enhance the immune response [adjuvants],” says the researcher.

He adds that the technology is usually divided into two components: the carrier particle (VLP), whose function is to make the immune system recognize the presence of a virus, and the viral antigen, responsible for stimulating the immune system to produce specific antibodies that prevent the pathogen from entering cells.
In case of the vaccine developed by the USP researchers, a platform already well studied by the scientists, called QβVLP, was used as VLP. It mimics the viral structure, allowing the immune system to “recognize” a threat. The antigen chosen was EDIII, a part of the Zika virus envelope protein whose function is to bind to a receptor on human cells.
“We inoculated the VLPs, produced in the USP laboratory by means of bacteria [Escherichia coli], chemically conjugated to the antigen. This combined structure mimics a real virus, with EDIII trapped on the outside of the platform”, describes Nelson Côrtes, a PhD student in Biotechnology and first author of the study. “When the formulation is injected into the body, this combination activates a strong immune system response, including antibodies and Th1-like cells, a subtype of T lymphocytes that plays crucial roles in the immune response.”
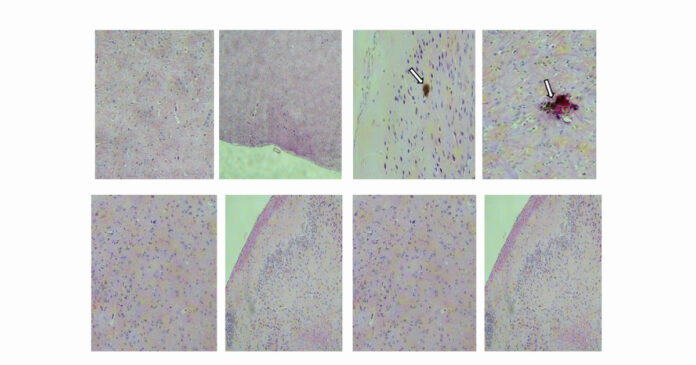
Tests carried out on genetically modified mice more susceptible to the virus showed that the vaccine induced the production of antibodies that neutralized the virus, did not allow the exacerbation of the infection and, consequently, the appearance of symptoms.
The researchers also investigated the effects of Zika virus infection on several organs in mice — including the brain, kidneys, liver, ovaries, and testes. “The vaccine demonstrated the ability to protect male mice from testicular damage,” says Côrtes. “This is significant given the known risk of sexual transmission of the Zika virus and its potential to impair testicular function, negatively affecting spermatogenesis and overall reproductive health,” the researcher adds.
Calibrated Aim
Zika virus has a particularity that makes the development of vaccines even more challenging: it is very similar to the four serotypes of dengue virus and co-circulates in the same transmission environment. The similarity causes antibodies to “confuse” one pathogen with another. It is what scientists call a cross-reaction, something that, at first, may even seem good – after all, the immune system recognizes a similar virus.
However, if the antibodies are not potent enough to prevent a second infection by another dengue serotype, for example, a boomerang effect occurs. The antibodies bind to the virus and make the host cell engulf the pathogen more easily. As a result, the organism itself helps the pathogen to infect the cells.
“The immunisation does not cause a cross-reaction, which is very positive. Previous studies by the group had already analyzed this issue and the use of the EDIII antigen allows the immune system to produce more specific antibodies to the Zika virus, avoiding the problem”, says Miranda.
The research received funding from the São Paulo Research Foundation (FAPESP) and involved researchers from USP’s Hospital das Clínicas (HC), Medical School (FM), Institute of Biomedical Sciences (ICB), School of Pharmaceutical Sciences (FCF), and Institute of Chemistry (IQ), as well as from the Federal University of São Paulo (Unifesp), the D’Or Research Institute, and the University of Pennsylvania, in the United States.
The article A VLPs based vaccine protects against Zika virus infection and prevents cerebral and testicular damage can be read here.
*By Maria Fernanda Ziegler, from Agência Fapesp. Adapted for Jornal da USP
English version: Nexus Traduções, edited by Denis Pacheco